.svg)
Optimize MSI testing
Rule-out MSS cases from the diagnostic workflow by screening with AI for digital pathology
As biomarker screening evolves, AI testing bridges the gaps
MSI status is predictive of response to immunotherapy across all solid tumors.1
In 2023, CRC was the second most common cause of death by cancer world-wide.2
Targeted therapies, including immunotherapies, are moving earlier in the treatment pathway, particularly into the neoadjuvant setting, guided by biomarker testing on biopsies.3
MSI, a genomic biomarker, represents ~11% of overall CRC population; 4 playing an important role in determining treatment.
Testing for a growing number of biomarker targeted therapies (HER2, FGFR2, Claudin 18.2) is depleting already limited biopsy tissue.5
The 5-year relative survival rate for GC drops sharply from 38% overall to just 8% in distant-stage disease, underscoring the importance of identifying actionable biomarkers earlier in disease progression.6
MSI status is among the five most frequently tested biomarkers in advanced GC stages used to refine the treatment plan.5
EC is the second most common gynaecological cancer diagnosed across the globe.
Pathological mechanisms and risk factors driving endometrial cancer are not yet fully understood — though research is beginning to close that gap.
The mortality rate is rising disproportionately among non-White ethnic groups, driven largely by unequal access to high-quality care.7
EC has the highest prevalence of MSI compared to all solid tumors; MSI status is key in determining diagnostic staging.8
Evolving guidelines are increasing the number of biomarkers to test for; expanding the diagnostic workflow burden for pathologists and clinicians.
Biopsied tissue is increasingly insufficient to meet the demands of multi-biomarker testing.
Product information
.svg)
Biopsy and surgical resection
Tested on more than 2500+ patients
50% rule-out with 0.99 NPV
70% rule-out with 0.98 NPV
.svg)
Biopsy
Tested on more than 230+ patients
62% rule-out with 0.99 NPV
.svg)
Biopsy and surgical resection
Tested on more than 230+ patients
21% rule-out with 0.96 NPV
32% rule-out with 0.97 NPV
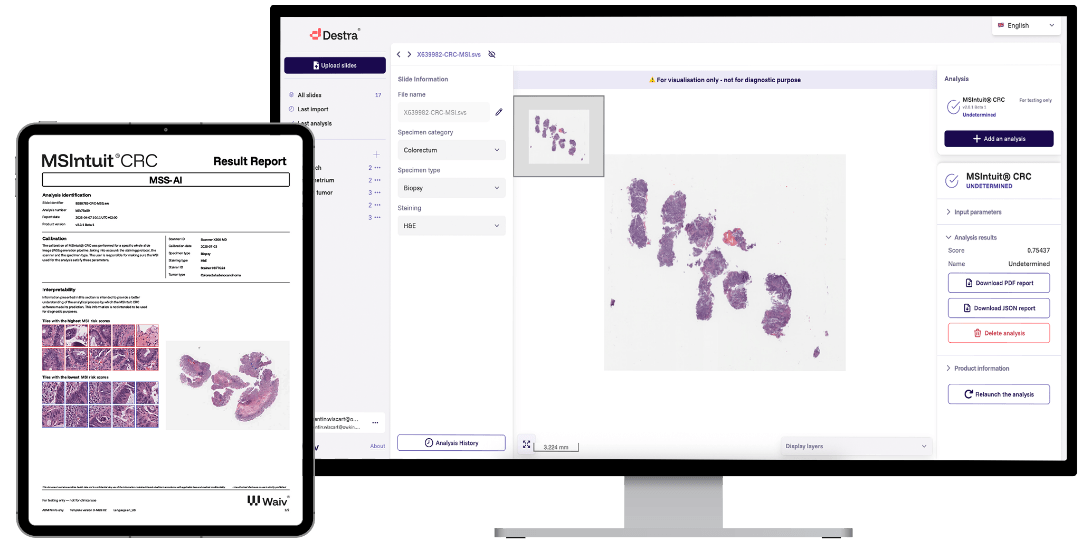
MSIntuit CRC demonstrates optimized MSI screening in real-world workflows9
MSIntuit Suite offers an enriched testing pool across Colorectal, Gastric, and Endometrial Cancers
Enabling centers to streamline their workflow, liberate auto-stainers and time for tasks with more added value and compensate for pathologist and technician shortages.
Interoperable precision test integration designed for existing workflows
The tissue sample is taken and prepared as H&E or HES slide.
The slide is scanned and digitized.

MSIntuit CRC deep neural networks extract and analyze features from the digitized slide.

MSS-AI
Patients can avoid additional testing thanks to high diagnosis precision.
Undetermined
Patients require further investigation to define microsatellite status with standard techniques.
Extensive clinical research and development underpin MSIntuit Suite’s robustness and applicability
Developed and rigorously tested with high-quality imaging data from varied clinical contexts and geographies.
MSIntuit Suite products have been deployed, tested, and evaluated in diverse clinical international settings.
Breakthrough image analysis and machine learning research form the bedrock of technological development for these products.
Citations
- Kawakami H, Zaanan A, Sinicrope FA. Microsatellite instability testing and its role in the management of colorectal cancer. Curr Treat Options Oncol (2015)
- World Health Organization. Colorectal cancer - WHO fact sheet, 2024
- https://dailynews.ascopubs.org/do/neoadjuvant-immunotherapy-msi-h-dmmr-colorectal-cancer
- Kang, YJ., O’Haire, S., Franchini, F. et al. A scoping review and meta-analysis on the prevalence of pan-tumour biomarkers (dMMR, MSI, high TMB) in different solid tumours. Sci Rep 12, 20495 (2022)
- https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf
- https://www.cancer.org/cancer/types/stomach-cancer/detection-diagnosis-staging/survival-rates.html
- https://www.cancer.org/cancer/types/endometrial-cancer/about/key-statistics.html
- https://ascopubs.org/doi/10.1200/PO.23.00118
- Organizational impact of an AI-based pre-screening tool for the diagnosis of MMR/MSI status in patients with colorectal cancer, Abstract #27 (Geppert, C. et al.), #28 (Levine, A. et al), #29 (Heukamp, L. et al.) at DP&AI 2025
- Results from MSIntuit CRC v2 performance validation published at USCAP 2025. A rule-out of 50% was obtained on biopsy samples, and 70% on surgical resection samples, 96% sensitivity on surgical resections, 95% sensitivity on biopsies, and 99% NPV for both surgical resections and biopsies, with 10% MSI prevalence.
Legal notice
MSIntuit® CRC v1 is CE-IVD marked for diagnostic use. MSIntuit® CRC v2 is a Class C (IVDR) medical device.
MSIntuit EC & MSIntuit GC are currently under development, and not for clinical use.
These devices are not FDA cleared or approved for use in the United States, and are available for Research Use Only.
For detailed information on proper use and precautions, please read carefully the instructions for use provided with the products. Images shown may represent the range of products, or be for illustration purposes only, and may not be an exact representation of the product.
MSIntuit EC & MSIntuit GC are developed within the PortrAIt consortium, a french consortium financed by the government within the framework of France 2030 and by the European Union - Next Generation EU within the framework of the France Relance Plan.
Manufacturer: Owkin Dx. Information updated on 15th April 2026 - Version W001V1
