.png)
Prognostic risk profiling for breast cancer
Next-generation AI testing from routine H&E to inform the treatment pathway for early breast cancer patients
Relapse risk assessment helps oncologists select the best treatment plan for patients
Incidence is high. Every 14 seconds, across the globe, a woman is diagnosed with breast cancer.4
Since 2008, worldwide breast cancer incidence has increased by more than 20 percent.4
Approximately 10% of all patients will relapse after their initial treatment.5
Nearly 70% of patients fall into the ER+/HER2- subgroup where there are a variety of treatments for oncologists to consider.
Those who relapse enter a chronic disease phase and are significantly more likely to have worse outcomes. It’s pivotal to identify these patients as early as possible to adapt their treatment strategies and evaluate their eligibility for treatment escalation, including newer targeted therapies, like CDK4/6 inhibitors1 2.
Identifying patients who are unlikely to relapse is also critical and a challenge. These patients may be able to safely avoid chemotherapies3, which often carry harsh side effects. But the risks attributed to relapse prompt oncologists to be conservative to avoid wrongly classifying high-risk patients, causing many to be potentially over-treated.
Current testing methods either lack consistency in accuracy6 7 8 do not address all subgroups effectively9 10 or they are expensive and not always accessible11, such as gene expression tests. The stakes are high for risk assessment in early breast cancer to limit the number of patients who are under or over treated.
RlapsRisk BC outperforms standard testing in accurately classifying patients as high or low risk in studies
International clinical studies show that RlapsRisk BC can more precisely stratify patients into high and low risk groups than both traditional clinical factors assessments and gene expression tests*.
In the pre-specified pooled analysis, It significantly separated patients into two groups of risk with an hazard ratio of 7.42 (95% CI, 4.32–12.75). With a consistently high Negative Predictive Value (NPV) across different cohorts (88% - 97.4%), RlapsRisk BC demonstrates it can also support safe identification of low risk patients. It also reclassifies those patients in the intermediate clinical risk category, and adds novel pathological based insights into the therapeutic decision making workflow12.
By adding additional information to the diagnostic workflow, used stand-alone or in combination with genomic signatures, RlapsRisk BC shows potential to help clinicians both reduce overtreatment and identify patients at higher risk who may benefit from adjuvant chemotherapy and other precision therapies12.
Integrating AI diagnostics seamlessly into the therapeutic and pathology workflows
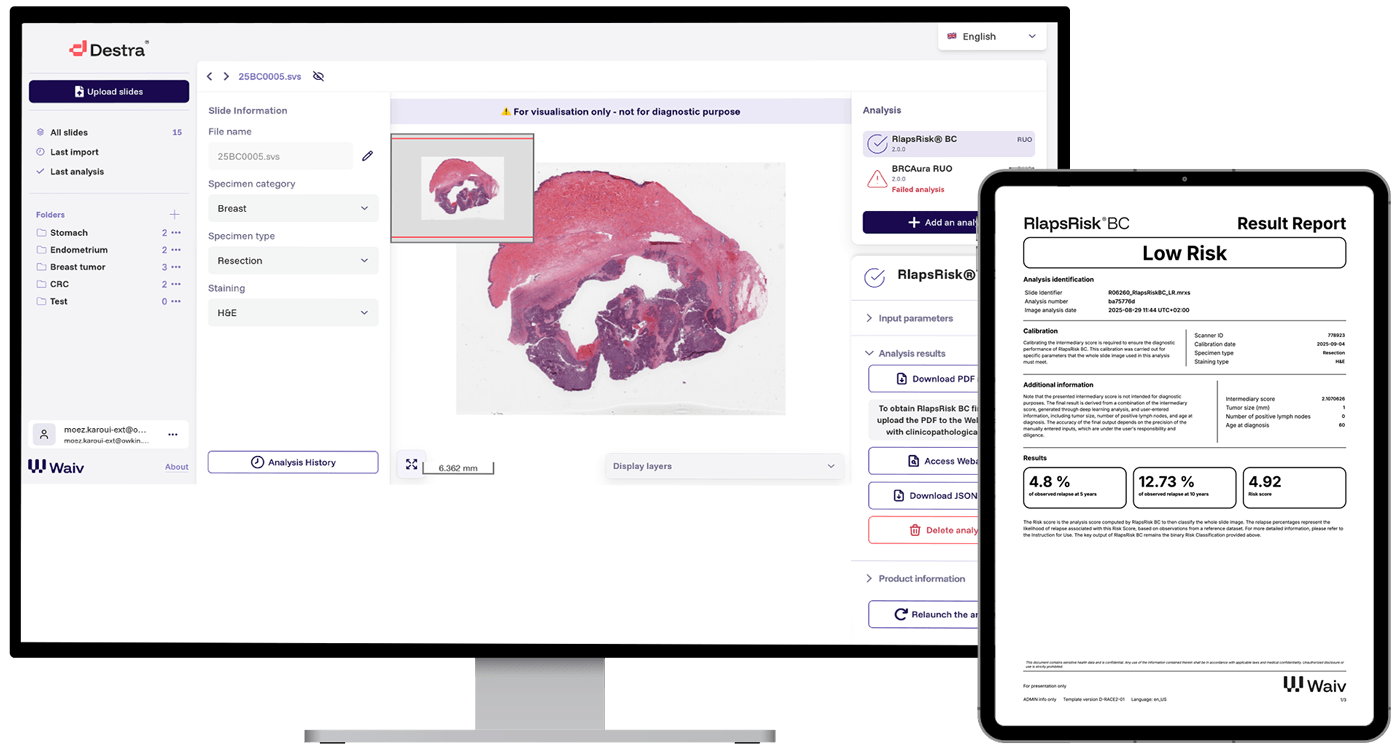
From resection tissue, the pathology lab prepares the H&E slides for diagnostic analysis.
After confirming ER+/HER2- status, the pathologist selects the slide that best represents the tumor for digitization and RlapsRisk BC analysis.

The slide is transformed into a whole slide image (WSI) via a slide scanner. The image is then available in the lab’s respective IMS or file management system.
The pathologist selects and verifies the image prior to triggering the AI analysis.

RlapsRisk BC’s deep neural networks extract and analyze features from the WSI, identifying which regions of the image are most prognostic of high risk and gives an initial report with a raw score.

Using an in-browser web app, the user supplies the patient’s age, number of invaded lymph nodes, and tumor size.
These readily available values are combined with the AI image analysis score to give the final risk classification.

RlapsRisk BC gives the result of either “high risk” or “low risk” via a pdf within the pathologists existing workspace.
The pathologist shares the final report with the oncologist or multidisciplinary tumor board to support therapeutic decisions.
RlapsRisk BC’s development and clinical studies set the foundation for product robustness and generalizability
Developed and evaluated with high-quality imaging data from varied clinical settings and geographic regions.
RlapsRisk BC and previous prototypes have been tested in various international lab settings, for three years.
Product development fostered in collaboration and informed by international breast oncology and pathology experts.
“Thanks to the solution we now have a better understanding of the underlying mechanism of highly aggressive tumors and the treatment needs for these patients. Identifying very high-risk patients earlier will enable us to adjust the therapeutic strategy for more favorable patient outcomes.”
Citations
- Mastro LD, Mansutti M, Bisagni G, Ponzone R, Durando A, Amaducci L, et al. Extended therapy with letrozole as adjuvant treatment of postmenopausal patients with early-stage breast cancer: a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 1 oct 2021;22(10):1458‑67
- Harbeck N, Rastogi P, Martin M, Tolaney SM, Shao ZM, Fasching PA, et al. Adjuvant abemaciclib combined with endocrine therapy for high-risk early breast cancer: updated efficacy and Ki-67 analysis from the monarchE study. Ann Oncol. 1 déc 2021;32(12):1571‑81
- Ferreira AR, Di Meglio A, Pistilli B, Gbenou AS, El-Mouhebb M, Dauchy S, et al. Differential impact of endocrine therapy and chemotherapy on quality of life of breast cancer survivors: a prospective patient-reported outcomes analysis. Ann Oncol Off J Eur Soc Med Oncol. 1 nov 2019;30(11):1784‑95
- Breast Cancer Statistics And Resources
- Long-term hazard of recurrence in HER2+ breast cancer patients untreated with anti-HER2 therapy, Strasser-Weippl et al. 2015, BMC.
- Gown AM. Current issues in ER and HER2 testing by IHC in breast cancer. Mod Pathol. 2008 May;21 Suppl 2:S8-S15. doi: 10.1038/modpathol.2008.34. PMID: 18437174
- Casterá C, Bernet L. HER2 immunohistochemistry inter-observer reproducibility in 205 cases of invasive breast carcinoma additionally tested by ISH. Ann Diagn Pathol. 2020 Apr;45:151451. doi: 10.1016/j.anndiagpath.2019.151451. Epub 2019 Dec 17. PMID: 31955049.
- Polley MY, Leung SC, McShane LM, et al. An international Ki67 reproducibility study. J Natl Cancer Inst. 2013 Dec 18;105(24):1897-906. doi: 10.1093/jnci/djt306
- Kalinsky K, Barlow WE, Gralow JR, et al. 21-Gene Assay to Inform Chemotherapy Benefit in Node-Positive Breast Cancer. New England Journal of Medicine (NEJM), 2021. DOI:10.1056/NEJMoa2108873
- Del Mastro L, Lambertini M, Pondé N, et al. Tailoring adjuvant chemotherapy and ovarian function suppression in premenopausal patients with HR+/HER2− early breast cancer: a critical review. Cancer Treatment Reviews, 2021. DOI: 10.1016/j.ctrv.2021.102010
- Blok EJ, Bastiaannet E, van den Hout WB, et al. Systematic review of the clinical and economic value of gene expression profiles for invasive early breast cancer available in Europe. Cancer Treat Rev. 2018 Jan;62:74-90. doi: 10.1016/j.ctrv.2017.10.012
- Manuscript under review with peer reviewed journal, and available as a pre-print on medRxiv: https://www.medrxiv.org/content/10.1101/2025.07.18.25331788v2
- Garberis, I., Gaury, V., Saillard, C. et al. Deep learning assessment of metastatic relapse risk from digitized breast cancer histological slides. Nat Commun 16, 5876 (2025)
Legal notice
RlapsRisk® BC is a Class C (IVDR) medical device.
This device not FDA cleared or approved for use in the United States, and is available for Research Use Only.
For detailed information on proper use and precautions, please read carefully the instructions for use provided with the products. Images shown may represent the range of products, or be for illustration purposes only, and may not be an exact representation of the product.
RlapsRisk BC is developed within the PortrAIt consortium, a french consortium financed by the government within the framework of France 2030 and by the European Union - Next Generation EU within the framework of the France Relance Plan.
Manufacturer: Owkin Dx. European Patent Application No. EP21306284.7 / International Application No. PCT/US2022/043692
Information updated on 22nd April 2026 - Version W002V2